IN Brief:
- EU MDR certification is now a gating item for new clinical electronics in Europe.
- Zenix pairs a large medical-grade touchscreen with workflow-focused software and feedback tools.
- Approval clears a path to broader deployment across EMS and hospital settings.
ZOLL Medical has received approval under the European Union’s Medical Device Regulation (MDR) for its Zenix professional monitor/defibrillator, a regulatory milestone that enables wider commercial deployment across European emergency medical services and hospital environments.
For electronics teams, MDR is not just a paperwork hurdle. The regulation tightens requirements around clinical evaluation, post-market surveillance, technical documentation, and quality systems, and it has materially increased the time and cost of bringing complex, software-heavy devices into the European market. Securing MDR approval signals that ZOLL’s hardware, software, and supporting documentation have cleared a more demanding bar than the previous Medical Device Directive regime.
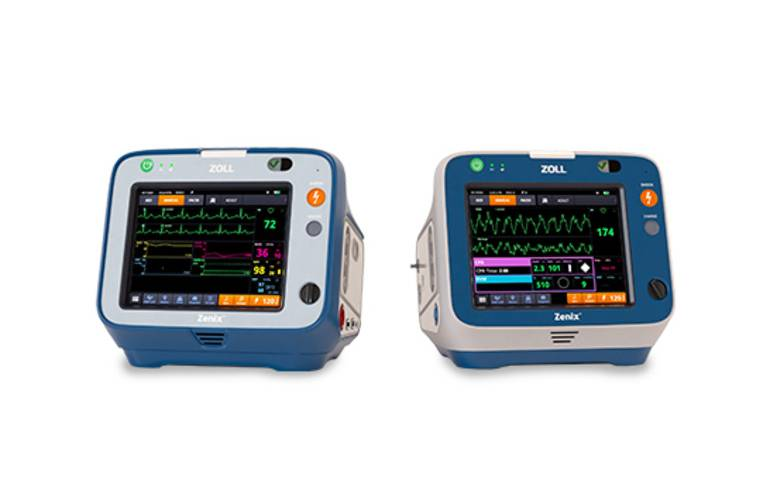
ZOLL positions Zenix as its most clinically advanced professional monitor/defibrillator, designed around a large, durable touchscreen and an interface intended to reduce friction under pressure. The device supports on-the-fly customisation, allowing clinicians to adjust views and settings in real time during resuscitation and transport, rather than stepping through fixed menus.
The platform also integrates ZOLL’s Real BVM Help and Real CPR Help technologies, providing real-time clinical feedback intended to improve ventilation quality and CPR delivery. That feedback loop is increasingly central to modern emergency electronics, where the device is expected to guide technique as well as deliver therapy, and where the user interface can be as safety-critical as the energy delivery path.
Elijah White, President of ZOLL Acute Care Technology, said, “Whether you are a hospital clinician or an EMS professional, the ZOLL Zenix monitor/defibrillator is designed to work the way you work,” adding that “virtually all functions of the device are accessible with three screen-touches or less.”
The approval arrives as EMS systems across Europe push for tighter integration between pre-hospital and hospital data flows, and as procurement teams scrutinise serviceability, fleet management, and software update pathways alongside core clinical functions. While ZOLL’s announcement centres on usability and clinical feedback, MDR compliance also implies a supporting structure for software maintenance and post-market obligations that can outlast a typical hardware refresh cycle.
ZOLL’s professional monitor/defibrillator range already spans multiple platforms, and Zenix now joins that portfolio as the company’s MDR-cleared option for European customers looking for a touchscreen-driven workflow with integrated feedback tools. The next test will be deployment at scale, where ruggedness, battery logistics, accessory supply, and software update discipline decide whether a device is merely approved or genuinely adopted.