IN Brief:
- FDA 510(k) clearance opens the US market for MARS Bioimaging’s point-of-care extremity scanner.
- The system uses photon-counting detector technology licensed from CERN’s Medipix3 collaboration to produce high-resolution 3D images with material differentiation.
- The clearance could widen access to premium musculoskeletal imaging beyond hospital radiology departments and into clinics, sports medicine settings, and ambulatory care.
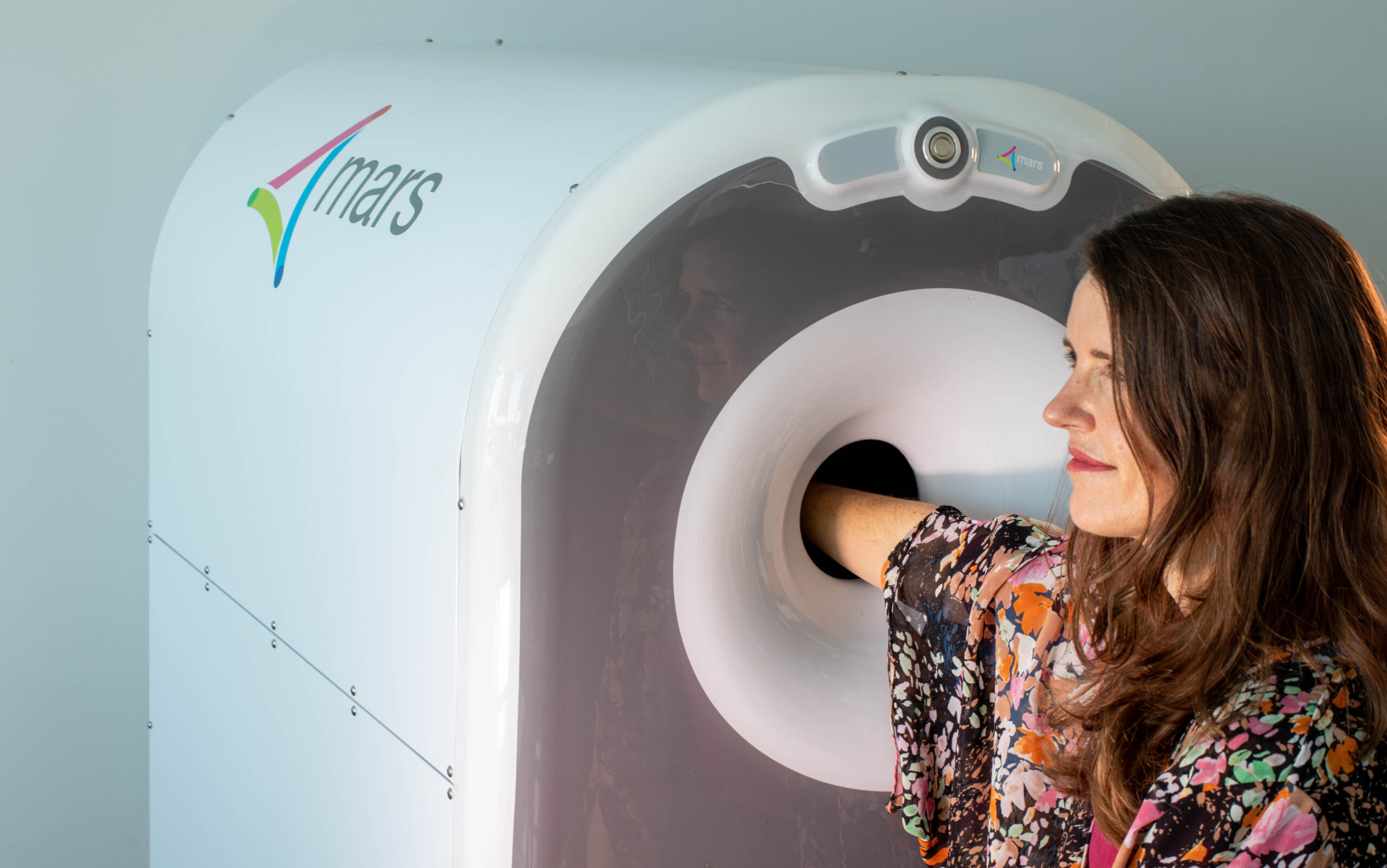
MARS Bioimaging has received US Food and Drug Administration 510(k) clearance for its portable photon-counting CT scanner for upper-extremity imaging, giving the New Zealand medical device company a formal route into the world’s largest CT market. The clearance covers a system designed for community and point-of-care settings rather than conventional hospital radiology departments, which is where most premium CT hardware has traditionally remained concentrated.
The company’s Extremity Scanner System is built to produce high-resolution 3D X-ray images with material differentiation across soft tissue, bone, blood vessels, and metallic implants. That combination matters in musculoskeletal imaging, where metal artefacts, small fracture detail, and the need to distinguish between adjacent materials can quickly determine whether a scanner is clinically useful or merely compact.
The technology stack is the more interesting part of the story. MARS is commercialising photon-counting detector technology derived from Medipix3, the CERN-developed platform originally created through particle physics research and later adapted for spectroscopic X-ray imaging. On its clinical product pages, the company describes the system as capable of capturing multiple X-ray energy levels in a single scan, allowing differentiation and quantification of intrinsic and extrinsic materials while operating at lower radiation dose than conventional approaches.
That technical promise has been building toward this point for several years. MARS had already been working with Hospital for Special Surgery in New York, and in 2024 the company said an HSS institutional review board had cleared its first US clinical trial. At the time, MARS pointed to an outpatient imaging model that could reduce the need to move patients through a main hospital radiology department for specialist wrist scans — a workflow issue that has long made advanced imaging slower, more expensive, and harder to access than the detector technology itself would suggest.
FDA clearance now turns that clinical and regulatory groundwork into a commercial one. MARS is targeting community clinics, sports medicine settings, ambulatory services, and office-based specialist care, with pre- and post-surgical planning, fracture healing assessment, and implant-related complication checks among the intended use cases. In effect, the company is trying to shift high-end CT capability closer to the point where orthopaedic decisions are actually being made.
For the wider electronics sector, the significance lies in where innovation is happening. Photon-counting CT is often framed as a scanner story, but the real differentiator sits in detector architecture, spectral processing, and the packaging of advanced sensing electronics into a system small enough to move beyond the imaging department. If that model takes hold, the next competitive argument in CT will not be size alone. It will be how much signal intelligence a compact detector platform can deliver without dragging hospital-scale infrastructure in behind it.